Biomedical 3D printing is revolutionizing the healthcare sector. According to a report from MarketsandMarkets, the biomedical 3D printing market is expected to reach $6.4 billion by 2026. This rapid growth showcases the incredible potential of this technology. From custom prosthetics to complex organ models, the applications are vast and varied.
Healthcare professionals are beginning to realize the value of personalized medical solutions. Custom implants and anatomical models can improve surgical outcomes and enhance patient safety. However, challenges remain in regulatory frameworks and material selection. The need for rigorous standards is critical for wider acceptance.
Moreover, ethical considerations are increasingly important in this field. As innovations emerge, so do concerns about accessibility and equity in healthcare. The path forward is not without hurdles, but biomedical 3D printing presents an exciting frontier for healthcare innovation. Addressing these challenges is essential for maximizing its potential.

Biomedical 3D printing is revolutionizing healthcare. This technology allows for the creation of patient-specific models and implants. According to a report by SmarTech Analysis, the biomedical 3D printing market is expected to exceed $3.5 billion by 2024. Innovations in bioprinting and materials science continue to drive this growth, enabling more complex and functional structures.
One common technique used is FDM (Fused Deposition Modeling), which involves layering materials to form a 3D object. This method is cost-effective and suitable for prototyping. SLS (Selective Laser Sintering) is another approach, offering higher precision and strength. While these techniques have advantages, they can also lead to challenges in material compatibility and structural integrity.
Tips: Consider the application when choosing a technique. Each method has specific material requirements. Collaborate with experts in biomedical engineering for optimal results. Regularly assess the printed models for imperfections, as small discrepancies can impact functionality. Understanding these nuances is vital for successful application in healthcare settings.
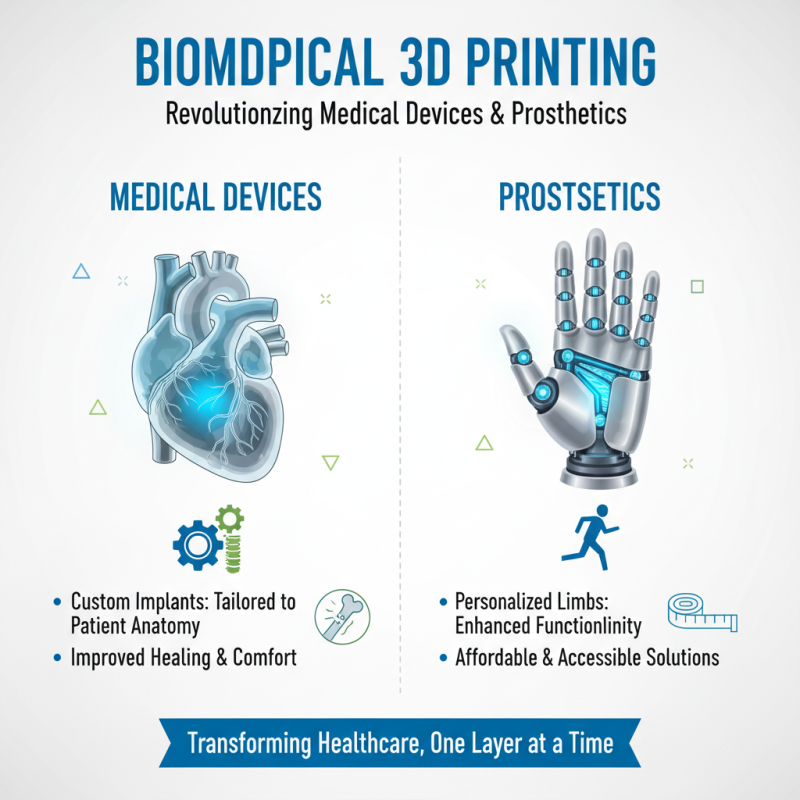
Biomedical 3D printing is transforming the medical field. One major application is in the creation of customized medical devices. These devices can be tailored to fit an individual’s specific anatomy. For example, custom implants are often more successful than standard options. They can improve healing times and patient comfort.
Prosthetics benefit greatly from this technology as well. Traditional prosthetics can be heavy and uncomfortable. 3D-printed prosthetics, however, can be lightweight and designed for comfort. This personalization ensures a better fit and expanded functionality. Additionally, various materials can be utilized, which enhances the user’s experience.
While these advancements are impressive, challenges remain. The regulatory landscape for 3D-printed medical devices is still developing. There are significant concerns about quality control and material safety. Healthcare professionals must approach these innovations with caution. Ensuring reliability is paramount. As the field evolves, continuous research and feedback from users will be essential.
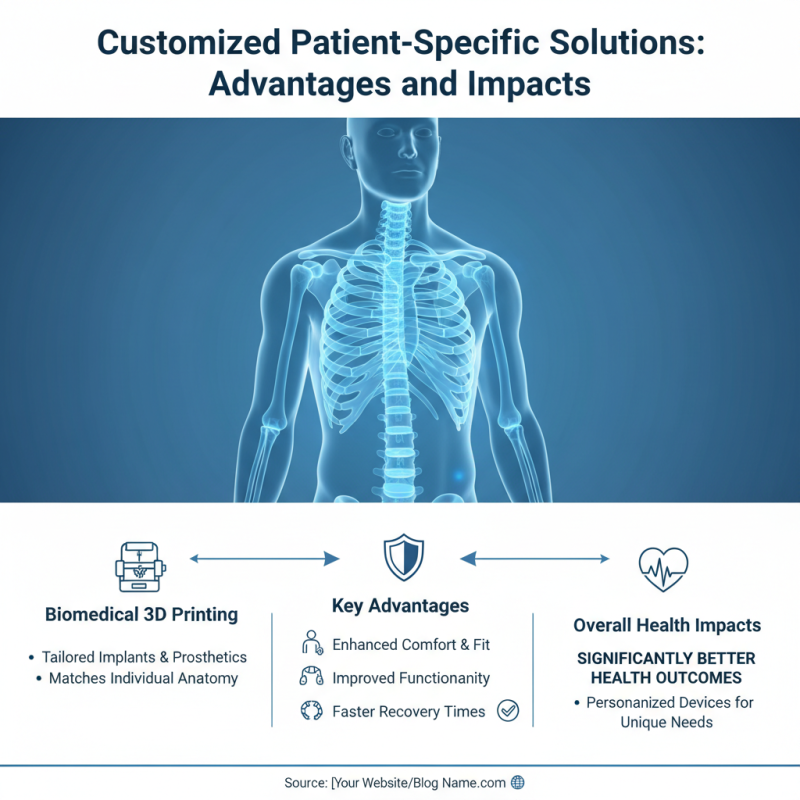
Biomedical 3D printing is transforming healthcare by offering customized patient-specific solutions. This technology allows healthcare professionals to create implants and prosthetics tailored to individual anatomical structures. For instance, a patient with a unique bone structure can receive a precisely fitted implant, enhancing comfort and functionality. Such personalized devices can significantly improve recovery times and overall health outcomes.
However, there are challenges associated with these innovations. The production process can be lengthy and resource-intensive. Moreover, the materials used may not yet be ideal for all applications. Ethical concerns also arise when considering the extent of customization possible. Balancing innovation with safety and efficacy remains a priority. Despite the obstacles, advancements continue to push the boundaries of what personalized medicine can achieve. Patient outcomes are already showing promise, but ongoing evaluation and improvement are crucial to fully realize the potential of 3D printing in healthcare.
The field of biomedical 3D printing is rapidly evolving, yet regulatory considerations remain a critical challenge. Regulatory bodies must address unique complexities presented by customized medical devices. Unlike mass-produced products, 3D-printed items can be tailored to individual patients. This variability complicates the assessment of safety and efficacy.
Quality assurance is essential. Companies must implement strict quality control processes. Documentation of each step in the printing and post-processing phases is vital. This ensures traceability and accountability. Regulatory frameworks are still catching up with technology. It can lead to confusion and delays in market entry. Companies often encounter hurdles related to premarket submissions and clinical evaluations.
There is also the need for ongoing collaboration between regulators and innovators. Open dialogue can help shape guidelines that are practical and supportive. Many professionals express concerns over the sufficiency of current regulations. Continuous adaptation and improvement of these standards are necessary. If done correctly, biomedical 3D printing can transform healthcare. The right regulations will foster innovation while ensuring patient safety.
Biomedical 3D printing is revolutionizing healthcare by providing tailored solutions to complex problems. One emerging trend is the creation of patient-specific implants. These implants are designed using imaging data from each patient, ensuring a perfect anatomical fit. This precision can lead to quicker recovery times and better patient outcomes. However, challenges remain in material biocompatibility, which can affect long-term success.
Another innovative application is the development of bioprinted tissues. Researchers are exploring ways to print functional tissues for research and regenerative medicine. For instance, creating skin grafts or organ models can aid in drug testing. Despite the progress, achieving full vascularization in 3D printed organs is still a significant hurdle. The technology continues to evolve, addressing complex biological requirements.
These advancements in biomedical 3D printing showcase the potential for improving healthcare. However, ethical considerations must also be addressed. The integration of this technology into clinical practice raises questions about regulation and access. Ensuring that innovations reach all patients equitably is vital for the future of healthcare.
This chart illustrates the growth in various applications of biomedical 3D printing in healthcare from 2020 to 2025, showcasing areas such as prosthetics, tissue engineering, and medical devices.
We use cookies to show you content that is relevant to you. By clicking “Accept”, you agree to the storing of cookies on your device to enhance site navigation, analyze site usage, and assist in our marketing efforts. Feel free to read more about cookies by clicking "settings"